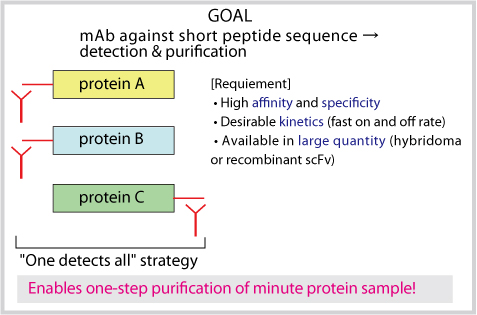
1. GOAL
Recombinant production of extracellular proteins in mammalian cells is routinely exercised in many labs around the globe including both basic and industrial researchers, but purification of target protein from the culture supernatant often demands method development/optimization dependent on individual project. We have developed a novel anti-peptide antibody P20.1 that can be used as both detection and affinity-purification tool suitable for the application in the recombinant production of human proteins. The intrinsic affinity of the mAb to the minimum epitope sequence (6aa) is low, allowing the mild elution from the affinity resin, and the total affinity can be increased by increasing the valency of either the tag or antibody. A protein purified using this system yielded diffraction-quality crystals that gave 1.5A resolution structure within 3 weeks. Three dimensional structure of P20.1-peptide complex and detailed information about sequence specificity are both available, opening the possibility for the further engineering of the system.